Potency Assay Services
We develop, qualify, and validate mechanism-driven potency assays for gene therapies and ATMPs across the entire development lifecycle.





We develop, qualify, and validate mechanism-driven potency assays for gene therapies and ATMPs across the entire development lifecycle.





Potency assays for gene therapies and ATMPs rarely behave ideally. Biological systems introduce variability that cannot be eliminated — only understood and managed. We design and execute potency assays that remain robust under real biological conditions:
Our approach ensures that potency readouts remain reliable, reproducible, and scientifically meaningful — even when biology is challenging.
With assays designed for regulator acceptance from day one.
With GMP constraints, robustness, statistical models and references standards all considered early in development.
Through early identification of biological variability and assay limitations — enabling robust development and a smoother progression.
We help clients anticipate biological and statistical challenges, and provide expert support when batch-to-batch variability or unexpected assay behavior occurs.
In the below example, cells were transduced across a range of MOI, and supernatants tested for protein activity (colorimetric assay).
Transduction signal increases with MOI, generating the characteristic sigmoidal dose–response shape comprising a lower asymptote, linear range, and upper asymptote. Reference and test samples overlay closely across the MOI range, demonstrating consistent assay performance and robust transduction.
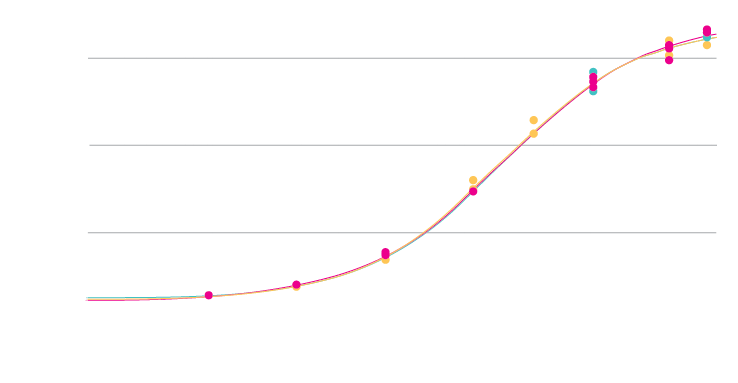
Triplicate measurements at each MOI demonstrate excellent precision, with low intra‑assay variability.
| MOI | Average % CV of triplicates |
|---|---|
| 8.40E+05 | 6.1 |
| 4.20E+05 | 6.0 |
| 1.05E+05 | 4.2 |
| 3.50E+04 | 4.4 |
| 1.17E+04 | 6.0 |
| 2.33E+03 | 10.0 |
| 4.67E+02 | 10.3 |
| 9.33E+01 | 14.9 |
Our validated assays typically demonstrate low intermediate precision variability, with operator‑to‑operator and day‑to‑day differences remaining tightly controlled
(Validation MOI 1.17E+04)
Highly trained operators with deep assay understanding and real-time troubleshooting capability. The same experts who develop the assay execute it under GMP — ensuring full continuity and no loss of knowledge.
Extensive experience in complex potency assays and systems with inherent biological variability.
Supported by advanced statistical expertise (full-curve and linear-range PLA, factor-to-reference approaches).
Deep regulatory experience across clinical phases enables phase-appropriate potency strategies and ensures GMP readiness from early development. Built on a developer mindset, with a clear understanding of program risk and long-term assay scalability.
Potency assay design grounded in the biology of your therapeutic, including:
Deliverable:
To ensure the assay is reliable and fit for early‑phase use.
Deliverables:
For later clinical phases and regulatory submission, we execute full method validation aligned with ICH guidelines.
Deliverable:
We support your overall potency assurance strategy, through assay implementation and ongoing commercial readiness with:

Please contact us using the form below.
"*" indicates required fields